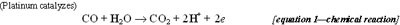Since electrochemical sensors work with the same theory but different materials, we have described only how the CO sensor works in this application note.
The carbon monoxide sensor consists of three electrodes immersed in a liquid electrolyte (a non-metalic liquid that conducts electricity, usually through acids or dissolved salts). The three electrodes are the working electrode, the reference electrode, and the counter electrode. The most important of these is the working electrode (WE). The working electrode is made of platinum, which is a catalytic metal to CO (it catalyzes the oxidation of CO to CO2), backed by a gas-permeable but hydrophobic (water-proof) membrane. The CO gas diffuses through the porous membrane and is electrochemically oxidized (see Equation 1: Chemical Reaction).
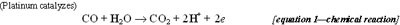 |
The electrons involved in the electrochemical reaction flow from the working electrode through the external circuit, producing the output signal of the sensor. In order for the reaction to take place, the thermodynamic potential of the working electrode is of critical importance. The reference electrode (RE) provides a stable electrochemical potential in the electrolyte. The reference electrode is protected from exposure to the CO gas, so that its thermodynamic potential is always the same and remains constant. Also, no current is allowed to flow through the reference electrode (this would change the thermodynamic potential.) A counter-electrode (CE) is provided to complete the circuit of the electrochemical cell. The counter electrode functions solely as the second half-cell, and allows electrons to enter or leave the electrolyte.
The circuit that controls the potential of the working electrode and converts the signal current to a voltage is called a potentiostat. A simplified diagram of the sensor and potentiostat is illustrated below. The signal (current) from the working electrode (WE) is converted to a voltage by operational amplifier U2. This circuit also maintains the voltage of the working electrode at the bias potential, Vbias. The reference electrode (RE) potential is compared to the stable input voltage, Vbias. The op-amp U1 generates a voltage at the counter electrode (CE) which is just sufficient to produce a current that is exactly equal and opposite to the working electrode current. At the same time, a constant voltage is maintained between the reference electrode and working electrode.
 |
The CO sensor is also equipped with a chemically selective filter. This filter removes potentially interfer-ing gases before they reach the working electrode. With the filter operating properly the sensor will have minimal response to interfering gases.
The technology described above can be modified to give sensors that will respond to the different gases of interest. This can be accomplished by using different working electrodes, bias potentials, and chemically selective filters. The following Table shows which catalysts are used for each gas.