The Importance of Vaccine Storage and Handling
Properly Storing Vaccines
Technology has had a huge impact on the effectiveness of vaccines. Thermometers have especially undergone the shift from consumer technology, simple digital home devices, to healthcare technology, information technologies used to enhance and optimize processes and improve care.
The earlier years of vaccine storage involved reading simple digital max/min thermometers that had to be checked at least twice a day with the temperatures manually being logged into a log book to be checked by authorities. The proper storage consisted of the practices listed below, which, for the most part, are still practiced today:
Vaccines must be stored in an environment with a controlled temperature, for instance a refrigerator that maintains a stable temperature ranging from 2˚C to 8˚C.
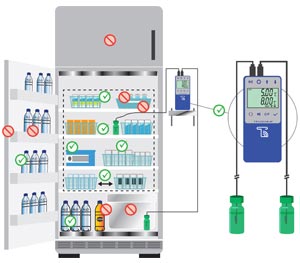
- Bottles of water should be placed on the door of the refrigerator to provide temperature stability, especially if the refrigerator door is left open
- Bins should be placed carefully in the refrigerator, take care to not place bins too close to refrigerator walls as walls could change temperature for temperature stability
- A thermometer, set to the out option and attached to the diluent vial, should then be taped in the center of the middle shelf
- Give the refrigerator time to reach the temperature within the cold chain range for vaccines to be properly stored and maintained
- One person, who understands how to handle these tools, should be chosen to oversee that storage and handling guidelines are met
- The temperature of the refrigerator holding the vaccines should be checked twice daily with maximum and minimum temperatures recorded in a log book
- The thermometer should be reset after each reading to ensure temperatures are read accurately
As more guidelines became apparent within the vaccine storing world, it became clear that there would have to be something  done to improve and facilitate the process of ensuring that the vaccines remained within the cold chain range. This caused the shift from min/max alarm thermometers to digital data-logging thermometers (DDLs). DDLs have an internal memory that logs the data regularly throughout the day along with an alarm should the temperature veer outside of required limits. This data can be downloaded for analysis and reporting needs. However, one concern with data logging was simply that someone could be away and therefore unable to hear the alarm sounding, as is often the case in practice, with facilities being unmanned over 50% of the time.
done to improve and facilitate the process of ensuring that the vaccines remained within the cold chain range. This caused the shift from min/max alarm thermometers to digital data-logging thermometers (DDLs). DDLs have an internal memory that logs the data regularly throughout the day along with an alarm should the temperature veer outside of required limits. This data can be downloaded for analysis and reporting needs. However, one concern with data logging was simply that someone could be away and therefore unable to hear the alarm sounding, as is often the case in practice, with facilities being unmanned over 50% of the time.
As guidelines and technology continued to improve, facilities began to request some change. Perhaps there could be a way to be alerted, if away at any point in time, for when the temperature of the refrigerator holding vaccines was acting awry. Therefore, simple solutions like TraceableLIVE® exist today. These cloud-based thermometers allow one to remain connected and alert on any personal device when out of the office or simply in a location where one cannot hear the alarm in case of worrisome temperature changes.
The storage process is far simpler now with the use of cloud-based monitoring thermometers causing a major increase in percentages of safe vaccines.
Properly Handling Vaccines
Another major problem lies in the mishandling of the vaccines as they are transferred from the manufacturer to the facility, which could put the vaccines at high risk for being exposed to temperatures that are not within the cold chain range. Like vaccine storage, the major concern of vaccine mishandling was ensuring that temperatures remained within the cold chain range. Immediate action and guidelines were provided by the The Centers for Disease Control and Prevention (CDC) as well for conducting proper handling practices.
Proper handling of vaccines is as such:
- An order should be placed for one month of vaccines at a time with an order request form
- A hard-walled cooler should be used to ensure that the vaccines are stable and remain cool while they are being transferred to a facility

- A frozen ice blanket should be placed at the bottom of the cooler and covered with one layer of bubble wrap to serve as a barrier between the ice and the probe
- Place the thermometer probe on top of and in the middle of the bubble wrap cover and place either a fridge cool water blanket on top of the thermometer or an ice blanket with a layer of bubble wrap beneath it on top of the thermometer
- Around 20 minutes should be given for the cooler to reach the cold chain range
- When the cold chain range is attained, vaccines are ready to be transported in the cooler
- When picking up vaccines, do not open boxes or bags that contain vaccines
- Place vaccines in same area as probe in the middle
- Transport cooler in seat beside your car to ensure that cold chain range is maintained
- Immediately transport vaccines from cooler to refrigerator and document/track them accordingly
Vaccines are now being properly stored and handled more than ever. This is the result of updated guidelines and consistent, reliable temperature monitoring. In the future, it is likely that many more vaccines manufactured will be fully potent and safe for public use due to strategic storing and handling.
Why Is Temperature Important?
Numerous aspects of humanity are dependent on temperature. It plays a crucial role in food, beverages, and agriculture. Our overall health is reliant upon temperature in many ways as well.
Vaccines are delicate biological substances that must be appropriately stored and handled from the time that they are manufactured until the time that they are used. With many vaccines, exposure to temperatures outside of the 2˚C to 8˚C range, otherwise known as the cold chain range, and even exposure to any light can cause a vaccine to lose its potency. Once a vaccine has lost its potency, the effects are irreversible.
The proper storage and handling of vaccines is imperative as patients depend on vaccines to prevent potentially dangerous illnesses. If mishandled whatsoever, the vaccines will inevitably lose potency costing up to thousands of dollars in revaccination and causing loss of patient confidence as they are put further at risk.
The CDC conducted a study that highlighted the improper storage and handling of vaccines. Unfortunately, 76 percent of the selected 45 providers in the study had some vaccines exposed to temperatures that were not within the cold chain range for some period. This study triggered the CDC to make changes and clarifications in guidance and requirements for vaccine care.
A Parting Reminder
When measuring for the temperature of vaccine storage devices, only calibrated temperature monitoring devices along with a certificate of calibration testing should be used. We have an ISO 17025 accredited calibration lab and calibrated lab equipment to ensure that all tools give accurate readings.
















